
KOH is available in pellet form typically with a purity of 98% (potassium carbonate is the usual impurity). Potassium hydroxide is also known as "caustic potash" and is a commonly used and relatively inexpensive chemical widely used in industry. 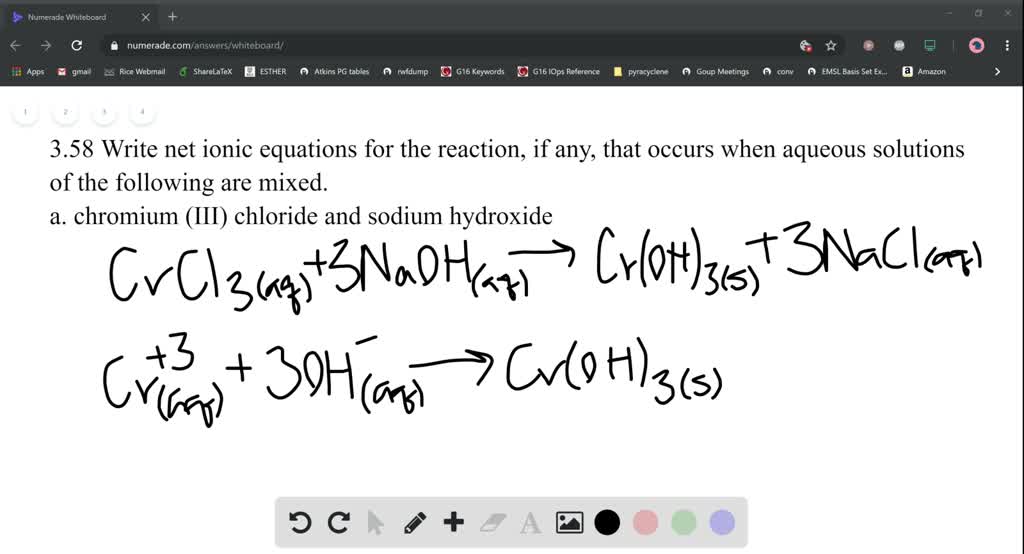
The synthesis process described here involves a reaction between ammonium nitrate and potassium hydroxide (KOH) that occurs in an aqueous solution. This webpage describes a process that is a relatively simple and safe way to make synthetic potassium nitrate that is quite pure and is perfectly suitable for use as a rocket propellant oxidizer. Ammonium nitrate cannot be used as a direct replacement for potassium nitrate in sugar propellant due to rapid decomposition that occurs at elevated temperature when in contact with organic materials such as sugars.Īs such, it is indeed fortuitous that potassium nitrate can be readily synthesized using ammonium nitrate as a base material. Although ammonium nitrate is potentially a very effective oxidizer, it is challenging to harness its capacity for a rocket propellant oxidizer due to its low combustion temperature and tendency to self-extinguish, drowning it its own water produced as the main product of combustion. Annual global consumption is around 20 million tonnes, and is found in most parts of the world for a wide variety of crops. On the other hand, ammonium nitrate (AN) is a common, inexpensive fertilizer (admittedly, AN is becoming less available due to restrictions on it sale). However, since this is a "specialized" fertilizer, its availability can be limited. The most economical form of potassium nitrate is 13-0-46 fertilizer. In some parts of the world, potassium nitrate can be hard to find, at least in economical quantities. Other Means of Potassium Nitrate Synthesis. 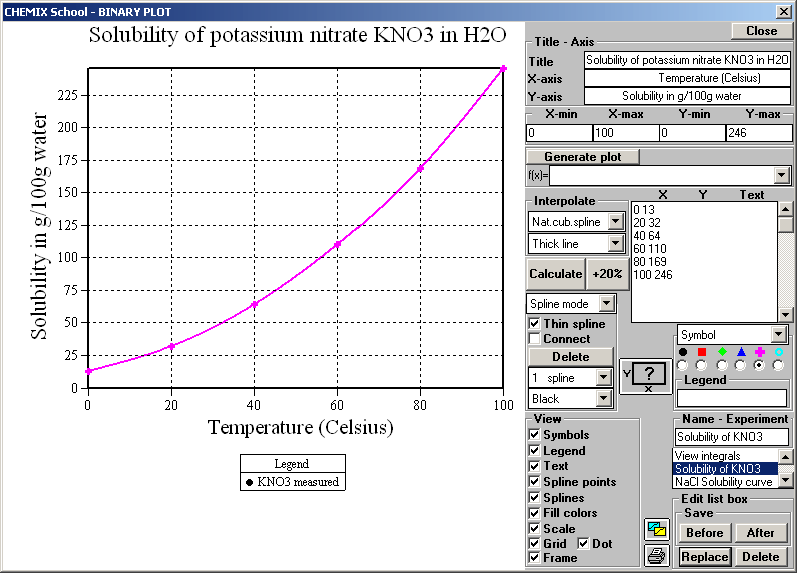
Synthesis of Potassium Nitrate from Other Chemicals Richard Nakka's Experimental Rocketry Web Site Richard Nakka's Experimental Rocketry Site
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
